Diagnosis of Gastrointestinal Stromal Tumor and Pathology Results
Gastrointestinal stromal tumor (GIST) is a cancer that may be suspected from its appearance in imaging techniques such as CT scans, but the diagnosis can only be determined by a pathologist after examining tumor tissue from a biopsy, or from the entire tumor after it has been surgically removed (resected). The main role of the pathologist is to determine the type of tumor (diagnosis). This is done by judging the characteristics of tumor cells viewed under a microscope and the results from a series of tests. The pathologist also gives an opinion regarding the prognosis (the risk of recurrence or metastasis).
|
Table 1. Tumor types in differential diagnosis with GIST |
|
leiomyoma |
|
Schwannoma |
|
neuroendocrine tumor |
|
fibromatosis or desmoid tumor |
|
angiosarcoma |
|
malignant mesothelioma |
|
dedifferentiated carcinoma |
|
metastatic melanoma |
Differential diagnosis is the process of identifying the tumor type from competing possibilities. Gastrointesstinal stromal tumor (GIST) became a clear cancer diagnostic category only in 1998, when it was discovered that nearly all GIST cells express KIT (a protein discussed further below), and that many GISTs show mutations in the KIT gene (Hirota et al, 1998). Prior to 1998 it was not certain that GISTs were fundamentally different from other abdominal neoplasms (cancers) in the broad category called mesenchymal tumors. Before the recognition of KIT mutations, GISTs were most often classified as leiomyoma, leiomyosarcoma, leiomyoblastoma, and gastrointestinal autonomic nerve tumor. Other mesenchymal tumors that comprise the differential diagnosis of GIST are included in Table 1.
Some of these tumors originate from the smooth muscle tissue of the GI tract (for example, leiomyoma and leiomyosarcoma) while others are of neural origin (for example, schwannoma and neuroendocrine tumor), and some such as fibromatosis or desmoid tumor are derived from connective tissue cells. The pathologist examines the appearance, shape, and features (morphology and histology) of tumor cells under the microscope to narrow the possibilities. The pathologist selects and applies immunohistochemical tests to tumor samples. These tests involve using antibodies to detect the presence of proteins that are expressed by the tumor cells. The pattern of proteins that are present or missing in a respective tumor in conjunction with the histomorphology of the tissue is taken into account for the final diagnosis.
For two recent reviews of GIST diagnosis and descriptions of pathology results for gastointestinals stromal tumor, see Parfitt et al (2006) and Miettinen and Lasota (2006). There is a special link to the latter paper on our site.
Morphology
GISTs do not all display the same cell morphology (shape and appearance of cells). About 70% of GISTs are composed of spindle cells (which look long and skinny), while about 20% are composed of epithelioid cells (which look round or polygonal), and the other 10% show mixed cells of both spindle and epithelioid types (pleomorphic). Illustrative slides of spindle and epithelioid cells are shown in Figure 1 and Figure 2 (greater magnification).
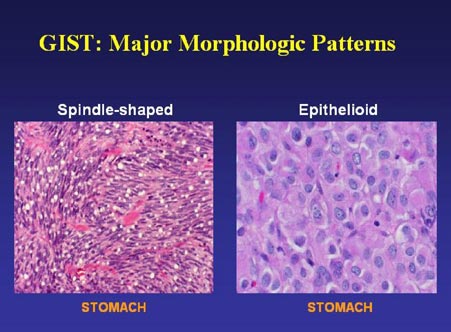
Figure 1. GIST cells of the spindle and epithelioid types (slide graciously provided by Cristina Antonescu, M.D.)
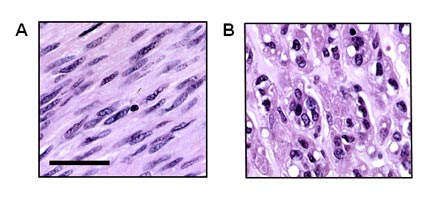
Figure 2. Gastrointestinal stromal tumors (GISTs) can show spindle cell or epithelioid histomorphology. A subset of tumors presents with a mixed phenotype. (A) GIST composed of neoplastic spindle cells. Note the blunt-ended nuclei that are reminiscent of smooth muscle cells. (B) GIST of the epithelioid type. H&E stains, scale bar indicates 50 µm. (Figure graciously provided by Anette Duensing, M.D.)
Immunohistochemistry
The best immunostaining method for identifying GIST is to test for expression of KIT, also called CD117. The KIT protein is a cell membrane-spanning signaling molecule (receptor tyrosine kinase) that normally functions to trigger cell growth when it is activated by its specific ligand stem cell factor (also see section on Mutation Analysis). KIT is normally expressed by the interstitial cells of Cajal (ICC), the “pacemaker cells of the gut” that send nerve signals to propel food along its course through the system via muscle contractions (peristalsis). KIT is also normally expressed by a few other types of cells in the body such as melanocytes in the skin, mast cells, and hematopoietic stem cells involved in making new blood cells (this is where “stem cell factor” got its name). With rare exceptions, KIT is not expressed by other abdominal tumors (Hornick and Fletcher, 2002; Hornick and Flechter, 2003). The presence and function of KIT in normal tissues is reviewed by Miettinen and Lasota (2005) and Roskoski (2005).
Researchers in Japan and in Sweden independently discovered in 1998 that GISTs express the KIT receptor protein (Hirota et al, 1998; Kindblom et al, 1998). Therefore, a positive result for KIT antibody testing is a strong indication of a GIST diagnosis. Figure 3 illustrates GIST tissue stained with KIT antibody.
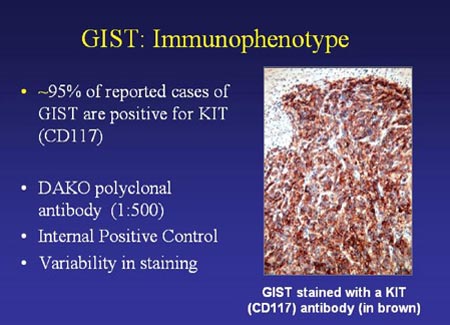
Figure 3. GIST cells stained with KIT antibody (slide graciously provided by Cristina Antonescu. M.D.)
Shown in Figure 4 is the contrast between KIT staining for only a few cells in normal gut wall versus staining for all the cells in a GIST.
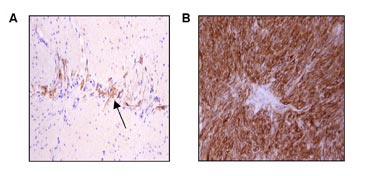
Figure 4. Interstitial cells of Cajal (ICCs) and gastrointestinal stromal tumors express the receptor tyrosine kinase KIT. (A) Immunohistochemical stain for KIT (CD117) in the Lamina muscularis of the gastrointestinal tract. Interstitial cells of Cajal (ICCs) within the myenteric plexus are highli
ghted by their KIT-positivity (arrow). (B) Uniform KIT positivity in a gastrointestinal stromal tumor. (Figure graciously provided by Anette Duensing, M.D.)
Figure 5 compares cells of leiomyosarcoma, the most frequent competing diagnosis, and spindle-cell GIST. Though the unstained samples appear similar, leiomyosarcoma cells do not stain for KIT. The discovery that GISTs are immunoreactive for KIT provided the first way to definitively separate GISTs from other tumors with similar appearance.
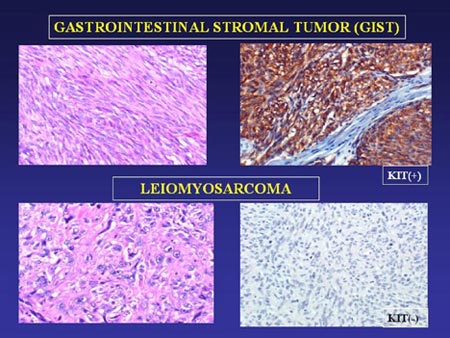
Figure 5. Comparison of GIST and Leiomyoma cells unstained (left-hand panels) and stained with KIT antibody (right-hand panels). (Slide graciously provided by Cristina Antonescu, M.D.)
Another immunohistochemical marker often found to be positive in GIST is CD34, a protein normally expressed by hematopoietic precursor cells (stem cells that generate new blood cells) and some interstitial cells of Cajal. Prior to the discovery of KIT, CD34 positivity was the best available indicator of a GIST diagnosis, but it was not very specific. Overall, about 60-70% of GISTs are positive for CD34, but this varies by tumor location. CD34 expression is highest in gastric GISTs (85%) but only about 50% of small intestinal GISTs express CD34 (Hasegawa et al, 2002; Rudolph, 2002). In less common GIST locations, Miettinen et al (2000) report CD34 positivity in 100% of esophageal GISTs, 65% of colonic GIST, 96% of rectal GISTs, and 65% of GISTs in non-GI locations such as mesentery and omentum. To sum this up, GISTs often stain for CD34, but this is neither required for GIST diagnosis nor unique to GISTs. Some competing diagnoses such as schwannoma and solitary fibrous tumor may stain for CD34.
Pathologists usually apply several other tests to identify the presence of markers indicating cell differentiation toward smooth-muscle or neural characteristics. A small percentage of GISTs may show positivity for smooth muscle actin (SMA), desmin, or S-100. This is not important if the GIST is KIT-positive. If the tumor is KIT-negative, then the pattern of results for these other markers will indicate if a non-GIST diagnosis is likely. Tumors of smooth muscle origin (leiomyoma and leiomyosarcoma [LMS]) are positive for SMA and desmin, while tumors of neural origin (schwannoma and others) are positive for S-100 and neuron-specific enolase.
KIT-negative GISTs are uncommon, but about 5% of GISTs do not stain for KIT although they otherwise resemble KIT-positive GISTs (Hornick and Fletcher, 2004; Medeiros et al, 2004; Orosz et al, 2005; Tzen et al, 2005). These cases present a challenge to the pathologist, who must evaluate the results of other tests, as well as the cell characteristics, to determine the most likely diagnosis. Additional tests to aid in this determination have been identified in the last few years. Protein kinase C theta (PKC theta) is a marker strongly expressed by GISTs, including KIT-negative GISTs (Duensing et al, 2004; Blay et al, 2004; Motegi et al, 2005; Kim et al, 2006). Staining for PKC theta is illustrated in Figure 6. In addition, a marker known as DOG1 (an abbreviation for “discovered on GIST 1”) is positive in GISTs (West et al, 2004). Positive results for these markers can differentiate a KIT-negative GIST from competing diagnoses. It is important to make the correct diagnosis to choose the correct treatment options.
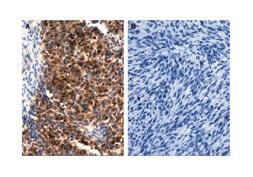
Figure 6. Protein kinase C theta (PKC theta) immunohistochemistry shows strong expression in a gastrointestinal stromal tumor (A) and no expression in a leiomyosarcoma (B). From Duensing et al , Cancer Research 2004; 64:5127-5131.
Proliferation Indicators
Cells reproduce or proliferate by dividing, and uncontrolled cell growth is one hallmark of cancer. The pathologist will usually provide some measure of how fast the tumor cells are dividing, as long as the sample is large enough to allow for this. Small samples from needle biopsies may not provide sufficient tissue, and the tissue in the biopsy sample may not be representative of the whole tumor. When a tumor is resected (surgically removed) the pathologist can examine the whole mass and select the best areas of the tumor for study. There are several ways to estimate proliferation. The higher the proliferation, the faster the tumor is growing and the more aggressively it can be expected to grow (if not resected) or to recur or metastasize (if removed).
Mitotic count entails visual examination of a set number of “high power fields” (HPF) under the microscope to count the number of tumor cells caught in the process of mitosis or cell division. “High power field” means one microscopic field of view at high magnification. The results are expressed as a ratio, such as 2/10 HPF (meaning 2 mitoses seen in 10 HPF) or 10/50 HPF (meaning 10 mitoses counted in 50 HPF). The higher the mitotic count, the faster the tumor is growing.
Other methods of measuring of how fast a tumor is growing are based on immunohistochemical stains for proteins, which are only present in proliferating (actively dividing) cells. Proliferating cell nuclear antigen (PCNA) and Ki-67 are the most common proteins looked at for this purpose. (Sometimes, the name “MIB-1” will be mentioned in this context. MIB-1 is actually the name of only one specific antibody directed against the Ki-67 protein. However, these two are sometimes used synonymously.) After staining for PCNA or Ki-67, the fraction of cells that are expressing these proteins and therefore thought to be actively dividing is counted by the pathologist. The result is given as a percentage. The higher this percentage, the faster the tumor is growing. This method seems to be more objective as compared to counting mitoses. However, there are some technical problems attached to it (e.g. variations in staining intensity) that pose a major downside to its use in everyday, routine diagnostics. These problems plus the additional time and costs are probably the main reasons why mitotic counts are still more widely used than immunohistochemical determination of proliferation, although the immunohistochemical methods are advocated by a number of authors.

