Sunitinib’s effectiveness against GIST and its side effects have been shown in several clinical trials. This page covers the following questions. Click a question to jump to the corresponding section.
What is the evidence of sunitinib effectiveness against GIST?
How long does it take to show a response to sunitinib?
Does mutation status affect response to sunitinib?
Does sunitinib keep working indefinitely?
What is the evidence of sunitinib effectiveness against GIST?
Sunitinib has been tested against GIST in a Phase I-II dose-finding and continuation trial, and later in a Phase III trial. There is also an ongoing worldwide treatment-use trial with a larger number of patients. The drug has been tested only in patients who either:
- did not respond to imatinib (Gleevec), or
- developed resistance to imatinib (Gleevec) and subsequently showed progression, or
- were intolerant of imatinib due to severe side effects.
Phase I-II data
In a Phase I dose-finding study summarized by Dr. Robert Maki at the 2005 ASCO meeting three dose schedules were evaluated, and the highest frequency of responses was shown on a 4-weeks-on/2-weeks-off schedule. Patients took 50 mg/day of sunitinib for 4 weeks, followed by 2 weeks without the drug (“washout period”). This schedule was then selected for use in the Phase II continuation study and in the Phase III study.
Patients who achieved partial response or stable disease for over 6 months in the Phase I study were eligible to continue into the Phase II study, also reported by Dr. Maki. The 32 patients Dr. Maki described in the continuation study comprise over half of the 55 patients in the Phase II study referred to as “Study B” in the sunitinib prescribing information and in the slides shown at www.sutent.com . The primary endoint of the Phase II study was objective response rate, as defined by RECIST. To meet the RECIST definition of “partial response” requires a decrease of 30% or more in the longest dimension of a single tumor or in the sum of the longest dimensions of a group of target lesions. Only 9.1% of all 55 patients in the Phase II trial demonstrated partial response. However, a greater proportion of patients achieved stable disease. Among the 32 Phase II patients described by Dr. Maki, about 20% of patients remained on treatment for over one year.
Mutation status and response. Dr. Maki’s presentation illustrated that the response of patients to sunitinib was related to the type of primary mutation shown in their GISTs. As shown in Figure 1, patients whose original (pre-imatinib) mutation status was exon-9-mutant or wild-type (no mutations in the KIT and PDGFRA genes) had a significantly longer duration of response than patients with exon-11 mutations. The median time to progression was 14.3 months for patints with original exon 9 mutations, 13.8 months for patients with wild-type KIT and PDGFRA, and 5.1 months for patients whose original mutation was in exon 11.
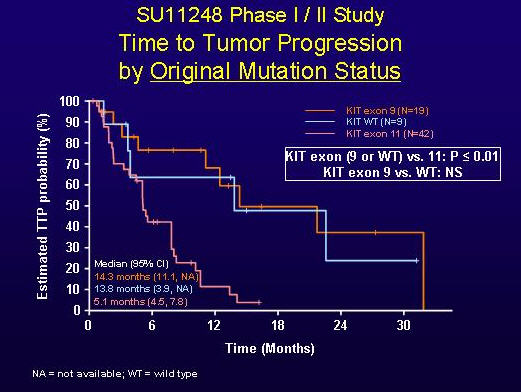
FIGURE 1. TIME TO TUMOR PROGRESSION BY ORIGINAL MUTATION STATUS (SLIDE GRACIOUSLY PROVIDED BY ROBERT MAKI, M.D., FROM PRESENTATION AT ASCO 2005, ABSTRACT 9011).
As shown below in Figure 2, this difference in response yielded a longer median overall survival time on treatment with sunitinib for patients whose original mutation status was wild-type (29.2 months) versus those with exon-11 mutations (12.7 months), and the median overall survival time had not yet been reached for the exon-9-mutant group at the time of the 2005 presentation.
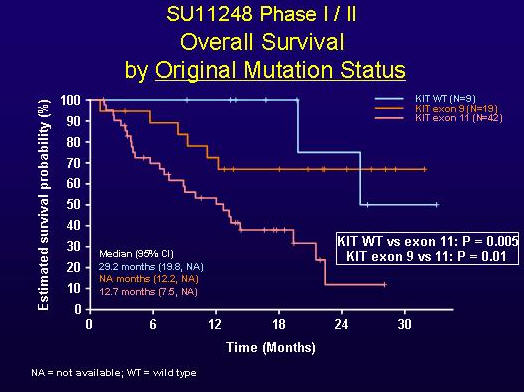
FIGURE 2. OVERALL SURVIVAL BY ORIGINAL MUTATION STATUS (SLIDE GRACIOUSLY PROVIDED BY ROBERT MAKI, M.D., FROM PRESENTATION AT ASCO 2005, ABSTRACT 9011).
Phase III trial sunitinib effectiveness data
Dr. George Demetri, principal investigator for the Phase III trial of sunitinib for GIST, presented an update on sunitinib (Sutent) effectiveness January 26, 2006 at the ASCO Gastrointestinal Cancer Symposium. The primary endoint of this study was time to progession (judged by tumor growth measured by RECIST). Because few patients had shown an objective response (tumor shrinkage constituting partial response by RECIST) in the Phase II study, the Phase III study was structured to include a placebo arm in which patients received substitute capsules rather than sunitinib. Patients were randomized into the treatment arm (207 patients receiving sunitinib) and the placebo arm (105 patients receiving dummy capsules). Individual patients in the placebo arm were able to cross over to the treatment arm when they showed progression by RECIST (>20% increase in tumor size). Patients were accrued from December 2003 until January 2005, when the first planned interim analysis showed a significant difference between arms in Time to Progression. The study was unblinded, and all patients then in the placebo arm were able to cross over to the treatment arm. As shown in Figure 3, the Time to Progression was significantly longer in the sunitinib arm (median = 27.3 weeks) than in the placebo arm (median = 6.4 weeks).
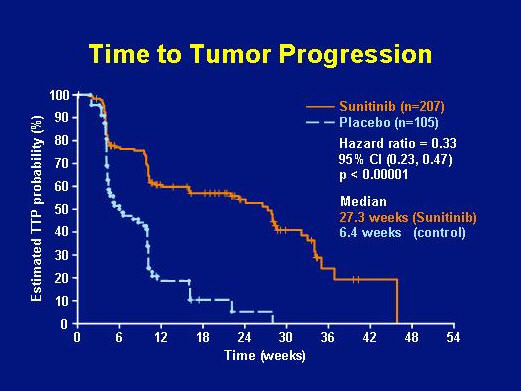
FIGURE 3. TIME TO TUMOR PROGRESSION BY TREATMENT ARM: SUNITINIB GROUP VERSUS PLACEBO GROUP (SLIDE GRACIOUSLY PROVIDED BY GEORGE DEMETRI, M.D., FROM PRESENTATION AT ASCO 2006 GI SYMPOSIUM).
Though the partial response rate for sunitinib in the Phase III trial was only 7%, an additional 19% of patients showed durable stable disease for over 6 months. The benefits of sunitinib were shown in all patient subsets (no response to imatinib, resistance to imatinib after a response, or intolerance of imatinib) and were independent of the dose of imatinib previously taken. The overall survival data for patients in the Phase III trial is shown as Figure 4.
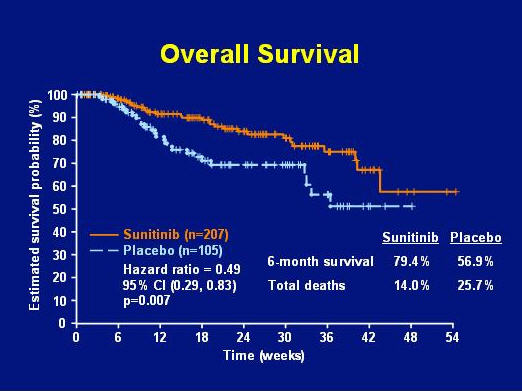
FIGURE 4. OVERALL SURVIVAL BY TREATMENT ARM: SUNITINIB GROUP VERSUS PLACEBO GROUP (SLIDE GRACIOUSLY PROVIDED BY GEORGE DEMETRI, M.D., FROM PRESENTATION AT ASCO 2006 GI SYMPOSIUM).
The formal publication of the Phase III clinical trial data for sunitinib against GIST appeared in October 2006 (Demetri et al, 2006). You can obtain the full paper from these sources:
- the health care professional section of the www.Sutent.com website, or
Most recently a new analysis was reported that accounted for the effect of the placebo and crossover design and provided a new estimate of survival. Click the title below to view the poster in Adobe Reader software, then use magnification control in Adobe Reader to enlarge and view one column at a time.
Long-term survival in a Phase III Trial of Sunitinib in imatinib-resistant/-intolerant GIST with Novel Statistical Analysis to Account for Crossover. Poster presented at November 2008 CTOS meeting by CR Garrett, X Huang, PG Casali, P Schoffski, ME Blackstein, M Shah, J Verweij, V Tassel, CM Baum, and GD Demetri.
The findings from this new statistical analysis were that, if the placebo group had not had to demonstrate progression before crossing over to treatment with sunitinib, then the overall survival results for all subjects would have been longer: “The long-term overall survival benefit of sunitinib relative to placebo was confirmed using RPSFT analysis. With this method the estimated median overall survival for patients on the sunitinib arm was twice that of patients on the placebo arm (73.9 weeks vs 35.7 weeks, with a hazard ratio = 0.469).”
Detailed Analysis of Survival and Safety with Sunitinib in a Worldwide Treatment-use Trial of Patients with Advanced Imatinib-resistant/intolerant Gastrointestinal Stromal Tumor. Poster presented at November 2008 CTOS meeting by B. Seddon, P. Reichardt, YK Kang, W. Ruka, A Nieto, A Breazna, T. Lechner, and GD Demetri.
The 2008 CTOS poster included data for 1127 patients enrolled in the ongoing worldwide trial. Median Time to Progression (TTP) was 41 weeks, and median Overall Survival (OS) was 75 weeks. This figure is very similar to the 73.9-week OS time for patients in the sunitinib arm of the Phase III trial. NOTE that mutation status information is not available for patients in this worldwide treatment-use trial. It is not known what the median TTP or OS data would be for those subjects most likely to benefit longest from sunitinib (exon-9-mutant and wildtype GIST patients).
Dr. Peter Reichardt commented about the sunitinib treatment-use trial in his piece Sunitinib (Sutent) Experience in Use on our Ask the Professional page. “Approximately 20% of patients have been stable for 2 or more years. Overall survival was longest for patients with age of <59 years, performance status ECOG 0-1, pretreatment with a maximum of 400 mg of imatinib or intolerance to imatinib. Worst overall survival was attributed to age above 60 years, poor performance status, pretreatment with higher doses of imatinib and primary resistance to imatinib.”
How long does it take to show a response to sunitinib?
Patients who switch from imatinib to sunitinib want to know as soon as possible whether the drug will work for them. Because clinical benefit of sunitinib is more likely to reflect stability than actual tumor shrinkage, CT scans will often indicate benefit only by lack of tumor growth. However, PET scans can indicate decreased tumor uptake of glucose tracer after only a few days of taking sunitinib, reflecting benefit.
Prior et al (2008) correlated PET results for GIST patients after 4 weeks of sunitinib therapy with their duration of Progression-Free Survival (PFS) on the drug. Those patients whose PET showed at least a 25% reduction in standard uptake value (SUV) had a longer PFS (median PFS = 29 weeks) than patients whose SUV remained within 25% of baseline (median PFS = 16 weeks). Patients whose SUV increased more than 25% above baseline after 4 weeks showed the shortest median PFS time of only 4 weeks. An absolute SUV of 8 g/ml or greater was also found to predict a shorter TTP.
For patients who need to make a decision with their oncologists about whether to stay on sunitinib or look for other drug options, PET scans can provide useful information.
Does mutation status affect response to sunitinib?
Sunitinib benefit is related to two different categories of mutations in patients:
- original mutation of the primary tumor
- secondary mutations in metastases or unresected primary tumors.
The Phase I-II and Phase III trial data discussed above demonstrated that the effectiveness of sunitinib varied for patients whose primary tumors show different original mutation types. Clinical benefit (partial response or stable disease) was significantly more common in patients with exon 9 mutations and patients without mutations of KIT or PDGRFA (wild-type KIT and PDGFRA) than in patients who originally had exon 11 KIT mutations. The occurrence of secondary mutations in the exon-11 group contributed to this difference. Secondary mutations may affect these areas:
- exon 13 or 14 (ATP and drug binding pocket area of KIT receptor)
- exon 17 or 18 (activation loop area of KIT receptor)
Heinrich et al (2008) published an analysis of mutation data for 78 of the 97 patients in the Phase I-II sunitinib trial, including both original mutation and secondary mutations present at the end of imatinib therapy. The greater clinical benefit of sunitinib for exon-9-mutation and wildtype patients occurs because these patients are unlikely to develop secondary mutations; only 19% of exon-9 patients developed secondary mutations, and none of the wildtype patients. In contrast, exon-11-mutant patients are prone to develop secondary mutations in the intracellular domains of the KIT receptor: 73% developed such mutations. Sunitinib was effective against tumors with secondary mutations in exon 13 or 14, but not against those with secondary mutations in exon 17 or 18.
Importantly, patients may harbor different secondary mutations in different metastatic lesions. Therefore, sunitinib may control some of a patient’s tumors, while others are unresponsive.
Dr. Ian Judson published an editorial [Journal of Clinical Oncology 26(33):5322-5325] titled Prognosis, Imatinib dose, and Benefit of Sunitinib in GIST: Knowing the Genotype about the impact of mutation status (also called genotype) on patients’ responses to imatinib and sunitinib. Link the title above to access this editorial summarizing these issues.
Does sunitinib keep working indefinitely?
The duration of clinical benefit from sunitinib is dependent upon the primary and secondary mutations shown by the patient’s original GIST and its metastases, as discussed above. Therefore, the duration of benefit also depends on mutation type. Patients with wildtype GIST (no mutations) and with KIT-exon-9-mutant GIST are less likely to develop secondary mutations, and therefore their benefit from sunitinib is longer-lasting. In contrast, patients with KIT-exon-11 mutations more frequently develop secondary mutations, some of which may be resistant to sunitinib. Because a single patient may have some metastases that can be controlled by sunitinib as well as some metastases with unresponsive secondary mutations, the duration of response may vary tumor by tumor within the same patient.
As stated by Dr. Peter Reichardt in his piece Sunitinib (Sutent) Experience in Use on our Ask the Professional page, “Approximately 20% of patients have been stable for 2 or more years. Overall survival was longest for patients with age of <59 years, performance status ECOG 0-1, pretreatment with a maximum of 400 mg of imatinib or intolerance to imatinib. Worst overall survival was attributed to age above 60 years, poor performance status, pretreatment with higher doses of imatinib and primary resistance to imatinib.” (This statement applies to patients in the worldwide treatment-use trial of non-randomized, unselected patients.)
Anecdotally, some patients have been stable on sunitinib for 5 years.

